The liver is the largest organ inside the body. It is located on the right side of the abdomen and protected by the ribcage. A person can survive with only a small portion of it in working order. Its functions include:
- destroying harmful substances, such as alcohol, and getting rid of waste products
- converting food containing fats and sugars to energy that is used by the body producing proteins in the blood
- producing bile to help with the digestion of food
Liver cancer can be a primary cancer (starts in the liver) or a secondary cancer (starts in another part of the body and spreads to the liver). Primary liver cancer is the sixth most frequently diagnosed cancer globally and the second leading cause of cancer death. Cancers can form from either the liver itself or from structures within the liver, including blood vessels, or the bile duct.
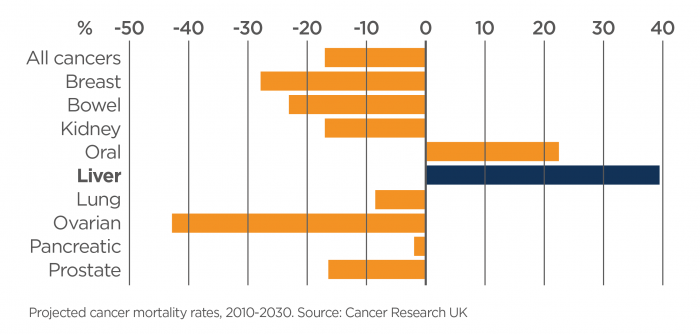
Liver disease is increasing due to lifestyle choices that result in obesity, diabetes, alcohol consumption that exceeds that recommended by the NHMRC and intravenous drug use that results in transmission of viruses that damage the liver such as Hepatitis B and C. Obesity causes what is called “fatty liver disease, or NASH”, and is leading to increased liver cancer in both men and women all across the world – in both developed and underdeveloped countries. In fact, liver cancer is increasing at an alarming rate globally and it is predicted that by 2030 it will have the worst outlook of all cancers.
Liver cancer is most often caused by cirrhosis (scarring of the liver tissue) by:
- excessive alcohol consumption
- long-term infection with a hepatitis virus – hepatitis B, C or D
- exposure to chemicals found in solvents, manufacture of plastics, herbicides
- smoking – hepatitis B or C infection increases the risk of liver cancer in people who smoke
- diabetes – people with diabetes have a risk that is two or three times higher than people who do not have diabetes
- haemochromatosis – an increased absorption of iron from the diet that causes liver damage
- drugs, including many antibiotics
There are very few nerves running through the liver and as such, there are often no noticeable symptoms in the early stages of liver cancer. In later stages, liver cancer can lead to an ache in the upper abdomen or back. Swelling, irritation or inflammation of the liver can result in pain in areas surrounding the liver. Other symptoms include loss of appetite, weakness and loss of weight (particularly loss of muscle in the arms and legs), nausea, fever and jaundice (yellowing of the skin and whites of the eyes). The abdomen may also be swollen.
Treatment for liver cancer will depend on whether it is a primary or secondary cancer. Treatment options may include:
- Surgery – to remove the cancer and damaged tissue. This is the main treatment for primary liver cancer. Surgery is only useful for secondary liver cancer if the cancer cells only affect one area of the liver.
- Chemotherapy – the use of tablets or injections of anti-cancer drugs. Sometimes, they are introduced into the veins via a drip, or injected directly into the artery that feeds the cancer in the liver. Chemotherapy is the main treatment for secondary liver cancer and it is also sometimes used for primary liver cancer.
- Radiotherapy – x-rays are used to target and kill cancer cells. Radiotherapy may help in treating some types of primary liver cancers, and may be used to relieve symptoms of pain and discomfort from secondary liver cancer.
The challenge facing liver cancer researchers is to find ways to enable early detection of liver damage that begins the process that leads to liver cancer. By the time a liver cancer is detected by conventional imaging methods, it is usually too large to remove surgically and/or has already spread to other parts of the body. Another challenge is to discover better ways to treat liver cancer because it is highly resistant to conventional anti-cancer drugs, consequently very few are approved by regulatory authorities abroad and in Australia. Finally, there is not enough known about the liver cancer cell; where it comes from and what makes it so difficult to kill.
Researchers in several laboratories at the Harry Perkins Institute are working collaboratively on all three strategies to achieve better outcomes for liver cancer patients. An approach towards early detection taken by Professor George Yeoh’s group is to focus on a liver progenitor/stem cell (LPSC). Normally, the damaged liver repairs itself by growing more liver cells. However, when it is badly affected in conditions that can lead to liver cancer, it calls on LPSCs to fix the damage. Hence the presence of these cells may be an early sign that liver cancer might follow. The Yeoh laboratory is developing methods to detect the LPSC in patients with liver disease; in their biopsies and in their blood.
Professor Peter Leedman’s laboratory is tackling the problem of discovering novel ways to kill liver cancer cells, amongst other research foci. A feature of these liver cancer cells is their ability to develop resistance to the most commonly used drug to treat liver cancer – Sorafenib. His laboratory has developed liver cancer cell lines that are resistant to Sorafenib and discovered underlying reasons for their resistance and is targeting these as a means to establish novel combination therapies. In addition, his team has been developing a new form of therapy for liver cancer that uses small RNAs to target the key growth pathways that drive the cancer. These small RNAs are called microRNAs or miRs and they represent a very promising way to tackle the disease. His team has discovered one miR, miR-7, is a very potent inhibitor of liver cancer cell growth and they are now developing miR-7 as a novel therapy for liver cancer with the aim of moving it into an early phase clinical trial in liver cancer patients. This would represent an example of “bench-to-bedside” translational research where the basic discovery is being translated to the patient via clinical trials.
Associate Professor Pilar Blancafort’s research group, which primarily focuses on breast cancer, is following a similar strategy but with a very different approach. Her group’s ability to alter the behaviour of breast and pancreatic cancer cells by modifying their genes is now directed at liver cancer cells. Cancerous LPSCs and HCs share many features that allow them to grow rapidly and resist death. The Blancafort laboratory has developed gene modifying tools that slow growth and render liver cancer cells susceptible to cancer drugs that cause cell death.
Equally important, Associate Professor Juliana Hamzah’s team have a novel approach to target liver cancer for diagnostic and therapeutic intervention. They have developed a molecular agent and a therapeutic agent that specifically recognise fibrous matrix and pro fibrotic cells in HCC. Treatment of preclinical models results in removal of fibrosis in HCC. They will now study how the anti-fibrotic agents affect HCC formation and progression in preclinical models as well as in human tissue biopsies.
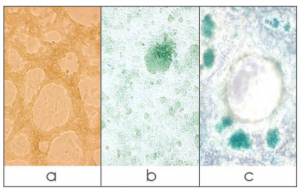
[Image]: BMEL TAT LPCs cell lines which have been differentiated to form ducts (a), hepatocytes (b) and ducts and hepatocytes (c). X-gal staining reveals LPCs expressing the beta-gal reporter which have differentiated into mature hepatocytes.
The Perkins is home to four additional research groups that are world leaders in their respective fields of cell biology who undertake liver cancer research collaboratively.
- Professor Alistair Forrest’s research group analyses the whole repertoire of genetic activity of different cell types and they have recently established the capacity to apply this to single cells. They are currently collaborating with the Yeoh laboratory to analyse LPSCs to distinguish the “good” LPSC that repair the liver from the “bad” LPSCs that lead to liver cancer.
- Professor Aleksandra Filipovska’s researchers are experts in developing pre-clinical models of metabolic diseases. They have established several models of liver fibrosis, liver steatosis and tumorigenesis as a consequence of energy dysfunction. In addition, the Filipovska group focuses on the development of compounds that can target cancer cells specifically and has previously used these to specifically target LPSC. This group is actively investigating the role of mitochondrial dysfunction in liver disease and devising treatments to treat liver cancer and steatosis.
- Professor Oliver Rackham’s group are experts at understanding the metabolic basis of disease at the cellular level and focus on the protein synthesis machinery inside mitochondria that is required for energy production. Protein synthesis defects are some of the most common causes of liver dysfunction in a range of genetically inherited metabolic diseases. Recently the Rackham group has defined a process by which the liver can be recovered via modulation of mitochondrial protein synthesis. These researchers will apply their expertise to understand the role of protein synthesis in LPSC that will underpin future strategies to treat liver cancer.
- The fourth group, under the leadership of Professor Ryan Lister, are world leaders in understanding the role of genetic modifications, specifically methylation alterations that regulate cell behaviour and especially in determining the fate of developing cells. This group has acquired and further refined tools that map these imprints in the genetic material of cells. The cancer atlas, a genetic blueprint of liver cancer cells shows many changes in genes that are caused by methylation and result in down-regulation of cancer protecting genes and up-regulation of cancer-causing genes. Their expertise will be invaluable once the LPSCs have been characterised and the distinguishing features of the “good” and the “bad” cell types are established.